Research shows that use of tobacco, alcohol, or illicit drugs or misuse of prescription drugs by pregnant women can have severe health consequences for infants. This is because many substances pass easily through the placenta, so substances that a pregnant woman takes also reach the fetus.91 Recent research shows that smoking tobacco or marijuana, taking prescription pain relievers, or using illegal drugs during pregnancy is associated with double or even triple the risk of stillbirth.92 Estimates suggest that about 5 percent of pregnant women use one or more addictive substances.93
Regular use of some drugs can cause neonatal abstinence syndrome (NAS), in which the baby goes through withdrawal upon birth. Most research in this area has focused on the effects of opioids (prescription pain relievers or heroin). However, data has shown that use of alcohol, barbiturates, benzodiazepines, and caffeine during pregnancy may also cause the infant to show withdrawal symptoms at birth.94 The type and severity of an infant's withdrawal symptoms depend on the drug(s) used, how long and how often the birth mother used, how her body breaks the drug down, and whether the infant was born full term or prematurely.95
Risks of Stillbirth from Substance Use in Pregnancy
- Tobacco use—1.8 to 2.8 times greater risk of stillbirth, with the highest risk found among the heaviest smokers
- Marijuana use—2.3 times greater risk of stillbirth
- Evidence of any stimulant, marijuana, or prescription pain reliever use—2.2 times greater risk of stillbirth
- Passive exposure to tobacco—2.1 times greater risk of stillbirth
Source: Tobacco, drug use in pregnancy, 2013
Symptoms of drug withdrawal in a newborn can develop immediately or up to 14 days after birth and can include94:
- blotchy skin coloring
- diarrhea
- excessive or high-pitched crying
- abnormal sucking reflex
- fever
- hyperactive reflexes
- increased muscle tone
- irritability
- poor feeding
- rapid breathing
- seizures
- sleep problems
- slow weight gain
- stuffy nose and sneezing
- sweating
- trembling
- vomiting
Effects of using some drugs could be long-term and possibly fatal to the baby:95
- birth defects
- low birth weight
- premature birth
- small head circumference
- sudden infant death syndrome (SIDS)
Risks of Sudden Infant Death (SIDS)
Children born to mothers who both drank and smoked beyond the first trimester of pregnancy have a twelvefold increased risk for sudden infant death syndrome (SIDS) compared to those unexposed or only exposed in the first trimester of pregnancy. New information from the NIH Safe Passage Study calls for stronger public health messaging regarding the dangers of drinking and smoking during pregnancy.
Illegal Drugs
- Marijuana (Cannabis)
More research needs to be done on how marijuana use during pregnancy could impact the health and development of infants, given changing policies about access to marijuana, significant increases in the number of pregnant women seeking substance use disorder treatment for marijuana use, and confounding effects of polysubstance use.96 A 2017 opinion posted by the American College of Obstetrics and Gynecology (ACOG) suggests that cannabis effects on fetal growth (e.g., low birth weight and length) may be more pronounced in women who consume marijuana frequently, especially in the first and second trimesters. ACOG recommends that pregnant women or women contemplating pregnancy should be encouraged to discontinue use of marijuana for medicinal purposes in favor of an alternative therapy for which there are better pregnancy-specific safety data.190
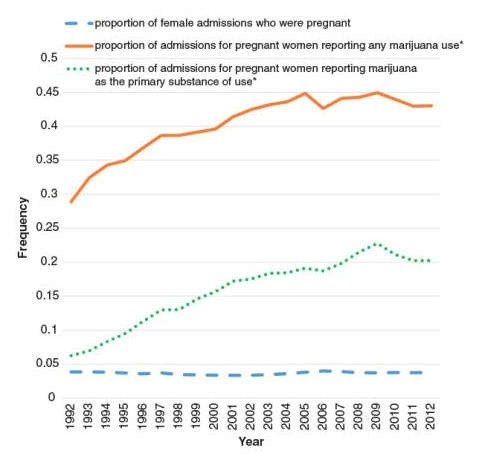
A recent study suggests that cannabis use more than doubled among pregnant women in the United States from 2010-1017. 191 Cannabis use was more common during the first trimester than during the second and third. Between 2002-2003 and 2016-2017, past-month cannabis use increased from 3.4% to 7.0% among pregnant women overall and from 5.7% to 12.1% during the first trimester. The study included information from 467,100 women aged 12-44 who participated in the National Survey on Drug Use and Health (NSDUH). Researchers also concluded that past-month clinician-recommended cannabis use was low among pregnant women, and nonmedical use was lower than among nonpregnant women, possibly reflecting the AGOC recommendations.
There is no human research connecting marijuana use to the chance of miscarriage,98,99 although animal studies indicate that the risk for miscarriage increases if marijuana is used early in pregnancy.100 Some associations have been found between marijuana use during pregnancy and future developmental and hyperactivity disorders in children.101–104 There is substantial evidence of a statistical association between marijuana smoking among pregnant women and low birth weight.105 Researchers theorize that elevated levels of carbon dioxide might restrict fetal growth in women who use marijuana during pregnancy.106 Evidence is mixed related to premature birth,107 although some evidence suggests long-term use may elevate these risks.108 Given the potential of marijuana to negatively impact the developing brain, the American College of Obstetricians and Gynecologists recommends that obstetrician-gynecologists counsel women against using marijuana while trying to get pregnant, during pregnancy, and while they are breastfeeding.109
Some women report using marijuana to treat severe nausea associated with their pregnancy;110,111 however, there is no research confirming that this is a safe practice, and it is generally not recommended. Women considering using medical marijuana while pregnant should not do so without checking with their health care provider. Animal studies have shown that moderate concentrations of THC, when administered to mothers while pregnant or nursing, could have long-lasting effects on the child, including increasing stress responsivity and abnormal patterns of social interactions.112 Animal studies also show learning deficits in prenatally exposed individuals.113,114
Human research has shown that some babies born to women who used marijuana during their pregnancies display altered responses to visual stimuli, increased trembling, and a high-pitched cry,115 which could indicate problems with neurological development.116 In school, marijuana-exposed children are more likely to show gaps in problem-solving skills, memory,117 and the ability to remain attentive.103 More research is needed, however, to disentangle marijuana-specific effects from those of other environmental factors that could be associated with a mother's marijuana use, such as an impoverished home environment or the mother's use of other drugs.118 Prenatal marijuana exposure is also associated with an increased likelihood of a person using marijuana as a young adult, even when other factors that influence drug use are considered.119 More information on marijuana use during pregnancy in NIDA's Marijuana Research Report. More research is needed, but for now, the Food and Drug Administration recommends that pregnant women should not use any vaping product, regardless of the substance.
Despite various surveys, the precise number of women who use marijuana while pregnant is unclear. One study found that women were about twice as likely to screen positive for marijuana use via a drug test than they state in self-reported measures. This suggests that self-reported rates of marijuana use in pregnant females is not an accurate measure of marijuana use and may be an underestimation.97
Very little is known about marijuana use and breastfeeding. One study suggests that moderate amounts of THC find their way into breast milk when a nursing mother uses marijuana.120 Some evidence shows that exposure to THC through breast milk in the first month of life could result in decreased motor development at 1 year of age.121 There have been no studies to determine if exposure to THC during nursing is linked to effects later in the child's life. With regular use, THC can accumulate in human breast milk to high concentrations.120 Because a baby's brain is still forming, THC consumed in breast milk could affect brain development. Given all these uncertainties, nursing mothers are discouraged from using marijuana.109,122 New mothers using medical marijuana should be vigilant about coordinating care between the doctor recommending their marijuana use and the pediatrician caring for their baby.
- Stimulants (Cocaine and Methamphetamine)
It is not completely known how a pregnant woman's cocaine use affects her child, since cocaine-using women are more likely to also use other drugs such as alcohol, to have poor nutrition, or to not seek prenatal care. All of these factors can affect a developing fetus, making it difficult to isolate the effects of cocaine.123
Research does show, however, that pregnant women who use cocaine are at higher risk for maternal migraines and seizures, premature membrane rupture, and placental abruption (separation of the placental lining from the uterus).93 Pregnancy is accompanied by normal cardiovascular changes, and cocaine use exacerbates these changes—sometimes leading to serious problems with high blood pressure (hypertensive crisis), spontaneous miscarriage, preterm labor, and difficult delivery.123 Babies born to mothers who use cocaine during pregnancy may also have low birth weight and smaller head circumferences, and are shorter in length than babies born to mothers who do not use cocaine. They also show symptoms of irritability, hyperactivity, tremors, high-pitched cry, and excessive sucking at birth.124 These symptoms may be due to the effects of cocaine itself, rather than withdrawal, since cocaine and its metabolites are still present in the baby's body up to 5 to 7 days after delivery.125,126 Estimates suggest that there are about 750,000 cocaine-exposed pregnancies every year.123
Pregnant women who use methamphetamine have a greater risk of preeclampsia (high blood pressure and possible organ damage),127 premature delivery, and placental abruption. Their babies are more likely to be smaller and to have low birth weight.128 In a large, longitudinal study of children prenatally exposed to methamphetamine, exposed children had increased emotional reactivity and anxiety/depression, were more withdrawn, had problems with attention, and showed cognitive problems that could lead to poorer academic outcomes.129,130
- MDMA (Ecstasy, Molly)
More research is needed on the effects of MDMA use during pregnancy. What research exists suggests that prenatal MDMA exposure may cause learning, memory,131 and motor problems in the baby.132,133
- Heroin
Heroin use during pregnancy can result in neonatal abstinence syndrome (NAS) specifically associated with opioid use. NAS occurs when heroin passes through the placenta to the fetus during pregnancy, causing the baby to become dependent on opioids. Symptoms include excessive crying, high-pitched cry, irritability, seizures, and gastrointestinal problems, among others.134
Medications
- Prescription and Over-the-Counter (OTC) Drugs
Pregnancy can be a confusing time for women facing many choices about legal drugs, like tobacco and alcohol, as well as prescription and over-the-counter (OTC) drugs that may affect the developing fetus. These are difficult issues for researchers to study because scientists cannot give potentially dangerous drugs to pregnant women. Here are some of the known facts about popular medications and pregnancy:
There are more than 6 million pregnancies in the United States every year,135 and about 9 out of 10 pregnant women take medication.136 The U.S. Food and Drug Administration issued rules on drug labeling to provide clearer instructions for pregnant and nursing women, including a summary of the risks of use during pregnancy and breastfeeding, a discussion of the data supporting the summary, and other information to help prescribers make safe decisions.137
See CDC Treating for Two webpageEven so, we know little about the effects of taking most medications during pregnancy. Fewer than 10% of prescriptions have enough information to determine fetal risks.138 This is because pregnant women are often not included in studies to determine safety of new medications before they come on the market.138 One study shows that use of short-acting prescription opioids such as oxycodone during pregnancy, especially when combined with tobacco and/or certain antidepressant medications, is associated with an increased likelihood of NAS in the infant.139
Although some prescription and OTC medications are safe to take during pregnancy, a pregnant woman should tell her doctor about all prescription and over-the-counter medications, and herbal or dietary supplements she is taking or planning to take. This will allow her doctor to weigh the risks and benefits of a medication during pregnancy. In some cases, the doctor may recommend the continued use of specific medications, even though they could have some impact on the fetus. Suddenly stopping the use of a medication may be more risky for both the mother and fetus than continuing to use the medication while under a doctor's care.140 This could also include medications to treat substance use disorders—something that is discussed in further detail in the "Sex Differences in Substance Use Disorder Treatment."
Some prescription and OTC medications are generally compatible with breastfeeding. Others, such as some anti-anxiety and antidepressant medications, have unknown effects,141 so mothers who are using these medications should consult with their doctor before breastfeeding. Nursing mothers should contact their infant's health care provider if their infants show any of these reactions to the breast milk: diarrhea, excessive crying, vomiting, skin rashes, loss of appetite, or sleepiness.142
Other Substances
- Alcohol
Alcohol use while pregnant can result in Fetal Alcohol Spectrum Disorders (FASD), a general term that includes Fetal Alcohol Syndrome, partial Fetal Alcohol Syndrome, alcohol-related disorders of brain development, and alcohol-related birth defects. These effects can last throughout life, causing difficulties with motor coordination, emotional control, schoolwork, socialization, and holding a job. More information can be found on the NIAAA Fetal Alcohol Exposure webpage.
Fetal alcohol exposure occurs when a woman drinks while pregnant. Alcohol can disrupt fetal development at any stage during a pregnancy—including at the earliest stages before a woman even knows she is pregnant.There is currently little research into how a nursing mother's alcohol use might affect her breastfed baby. What science suggests is that, contrary to folklore, alcohol does not increase a nursing mother's milk production, and it may disrupt the breastfed child's sleep cycle.143 The American Academy of Pediatrics recommends that alcohol drinking should be minimized during the months a woman nurses and daily intake limited to no more than 2 ounces of liquor, 8 ounces of wine, or two average beers for a 130-pound woman. In this case, nursing should take place at least 2 hours after drinking to allow the alcohol to be reduced or eliminated from the mother's body and milk. This will minimize the amount of alcohol passed to the baby.144
- Nicotine (Tobacco Products and e-Cigarettes)
Almost 10 percent of pregnant women in the United States have smoked cigarettes in the past month.13 Carbon monoxide and nicotine from tobacco smoke may interfere with the oxygen supply to the fetus. Nicotine also readily crosses the placenta, and concentrations of this drug in the blood of the fetus can be as much as 15 percent higher than in the mother.145 Smoking during pregnancy increases the risk for certain birth defects, premature birth, miscarriage, and low birth weight and is estimated to have caused more than 1,000 infant deaths each year.146 Newborns of smoking mothers also show signs of stress and drug withdrawal consistent with what has been reported in infants exposed to other drugs. In some cases, smoking during pregnancy may be associated with sudden infant death syndrome (SIDS), as well as learning and behavioral problems and an increased risk of obesity in children. In addition, smoking more than one pack a day during pregnancy nearly doubles the risk that the affected child will become addicted to tobacco if that child starts smoking.147 Even a mother's secondhand exposure to cigarette smoke can cause problems; such exposure is associated with premature birth and low birth weight, for example.148
Research provides strong support that nicotine is a gateway drug, making the brain more sensitive to the effects of other drugs such as cocaine.149 This shows that pregnant women who use nicotine may be affecting their fetus's brain in ways they may not anticipate. Additionally, e-cigarettes (or e-vaporizers) frequently contain nicotine. Therefore, those products may also pose a risk to the fetus's health. More research is needed, but for now, The Food and Drug Administration recommends that pregnant women should not use any vaping product, regardless of the substance.
Similar to pregnant women, nursing mothers are also advised against using tobacco. New mothers who smoke should be aware that nicotine is passed through breast milk,150 so tobacco use can impact the infant's brain and body development—even if the mother never smokes near the baby. There is also evidence that the milk of mothers who smoke smells and may taste like cigarettes. It is unclear whether this will make it more likely that exposed children may find tobacco flavors/smells more appealing later in life.151
Secondhand Smoke
Newborns exposed to secondhand smoke are at greater risk for SIDS, respiratory illnesses (asthma, respiratory infections, and bronchitis), ear infections,88 cavities,152 and increased medical visits and hospitalizations.153 If a woman smokes and is planning a pregnancy, the ideal time to seek smoking cessation help is before she becomes pregnant.