NIDA Points of Interest
About NIDA
Advancing the science on drug use and addiction
Director's Page
Nora D. Volkow, M.D., became Director of NIDA in May 2003.
Grants & Funding
Research grants, contracts, and supplements related to drug use and addiction.
NIDAMED: Clinical Resources
Substance use screening tools, guidelines, and other resources.
Research & Training
Programs to support research training from high school through tenure.
NIDA Research Programs & Activities
Learn about NIDA-supported research and cross-agency research activities on drug use and addiction.
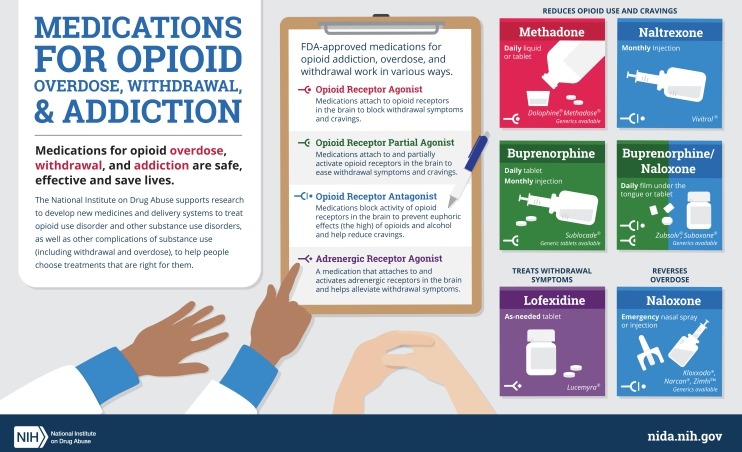
Medications for Opioid Use Disorder
Medications for opioid use disorder are safe, effective, and save lives. This NIDA-produced video takes a closer look at these medications and how they work.
Explore Topics in Substance Use and Addiction Science
Upcoming Meetings/Events
National Advisory Council on Drug Abuse: Open Session – September 2025
|
Neuroscience Center, 6001 Executive Blvd
National Advisory Council on Drug Abuse: Open Session – February 2026
|
Neuroscience Center, 6001 Executive Blvd