This research shows:
- An experimental compound with a dual action at two opioid receptors may provide powerful pain relief without many of opioids' harmful side effects.
- The compound may also have potential as a treatment for opioid addiction.
A novel compound represents a potential advance toward the goal of nonaddicting analgesics that are at least as effective as opioids but without typical opioid liabilities. The new compound, called AT-121, may also have potential as a treatment alternative for opioid addiction.
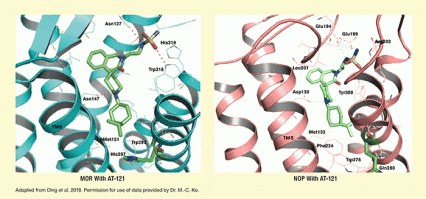
Dr. Nurulain Zaveri and colleagues at Astraea Therapeutics used medicinal chemistry, computer modeling, and structure-based drug design to design and develop AT-121. Like opioids such as morphine and oxycodone, AT-121 binds to the mu opioid receptor (MOR). However, unlike those opioids, AT-121 also binds to another opioid receptor called the nociceptin/orphanin FQ peptide (NOP) receptor (see Figure 1). According to Dr. Zaveri, this interaction with the NOP receptor enhances AT-121’s analgesic effect and blocks unwanted side effects often seen with current opioid medications.
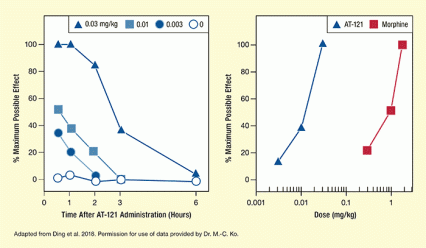
Dr. Huiping Ding, Dr. Mei-Chuan Ko, and colleagues at Wake Forest School of Medicine conducted preclinical tests of AT-121. After confirming in vitro that AT-121 bound strongly to and activated both MOR and NOP receptors, the research team assessed its pain-suppressing efficacy. In one experiment, the researchers found that AT-121 at a dose of 0.03 mg/kg completely suppressed monkeys’ discomfort when their tails were placed in very warm (50 degrees C) water. To achieve the same level of pain suppression with morphine, a dose 100 times higher was required (see Figure 2). In another experiment, AT-121 reduced heightened pain sensations like those that are sparked by hypersensitive pain fibers in some chronic pain conditions. Moreover, monkeys were no more likely to self-administer AT-121 than saline, indicating that the compound was not reinforcing, and therefore unlikely to motivate misuse.
The researchers also established that AT-121 may have potential as a treatment for opioid addiction. Monkeys that were pretreated with AT-121 before being exposed to oxycodone exhibited no signs of experiencing oxycodone reinforcement.
In monkeys, even very high doses of AT-121 produced none of the harmful side effects associated with opioid drugs, such as altered respiration, heart rate, blood pressure, or body temperature; sedation; or motor impairment. Also, unlike traditional opioids, repeated treatment with AT-121 did not produce physical dependence, tolerance, or excessive pain sensitivity, which can occur with long-term opioid treatment.
In earlier research, Dr. Ko and colleagues identified a buprenorphine-like compound, BU08028, that acts on all four opioid receptors to deliver powerful, non-reinforcing pain relief. However, AT-121’s targeted activity at only the NOP and MOR receptors yielded the added advantage of an attractive profile for treatment of opioid use disorders.
Dr. Zaveri says, "A single bifunctional compound with activity at two targets, such as AT-121, is a better pharmaceutical strategy than combining different compounds, which may have different pharmacokinetic properties, rates of metabolism, and propensities for drug–drug interactions." Dr. Zaveri and colleagues are now conducting toxicology and safety tests of AT-121, after which they hope to proceed to clinical trials.
This study was supported by NIH grants DA032568, DA027811, DA042465, DA040104, and DA044775.
- Text Description of Figure 1
-
The figure shows how AT-121 interacts with the mu opioid receptor (MOR) and the nociceptin/orphanin FQ peptide (NOP) receptor. In the left panel, teal and gray coils with lightly teal linear and ring structures indicate the MOR. The green structure with several rings as well as blue, red, and yellow arms indicates an AT-121 molecule. Dotted lines indicate where AT-121 atoms interact with specific amino acids in the MOR. In the right panel, pink and gray coils with lightly pink linear and ring structures represent the NOP receptor. The green structure with several rings as well as blue, red, and yellow arms again indicates an AT-121 molecule. Dotted lines indicate where AT-121 atoms interact with specific amino acids in the NOP.
- Text Description of Figure 2
-
The two graphs illustrate pain suppression and potency of AT-121 in rhesus monkeys. The left panel shows the effectiveness of three different doses of AT-121 in relieving pain caused by immersion of the monkey’s tails in warm water at specific time points after injection. The horizontal x-axis shows time after AT-121 administration in hours from 0 to 6 hours. The vertical y-axis shows the percent of maximum possible effect, indicating the percentage of a maximum time interval that the animals left their tails in the warm water. Pain suppression was measured at 0.5, 1, 2, 3, and 6 hours after injection. In control animals receiving no AT-121 (curve with open circles), the percentage of maximum possible effects was about 0 percent at all five time points. In animals receiving 0.003 mg/kg AT-121 (curve with filled circles), duration of pain suppression was about 35 percent of maximum possible effect at 0.5 hours, about 20 percent at 1 hour, about 4 percent at 2 hours, and about 0 percent at 3 hours and 6 hours. In animals that received 0.01 mg/kg At-121 (curve with filled blue squares), duration of pain suppression was about 50 percent of maximum possible effect at 0.5 hours, about 37 percent at 1 hour, about 20 percent at 2 hours, and 0 percent at 3 hours and 6 hours. For animals receiving 0.03 mg/kg AT-121 (curve with filled blue triangles), duration of pain suppression was 100 percent of maximum possible effect at 0.5 hours and 1 hour, about 85 percent at 2 hours, about 37 percent at 3 hours, and about 5 percent at 6 hours.
The right curve compares the effectiveness of different doses of AT-121 and morphine in relieving pain. The horizontal x-axis shows the dose of the agents used on a logarithmic scale from 0.001 to 2. The vertical y-axis shows the percent of maximum possible effect, indicating the percentage of a maximum time interval that the animals left their tails in warm water. For AT-121 (indicated by the blue curve), duration of pain relief with a dose of 0.003 mg/kg was about 15 percent of the maximum possible effect, with a dose of 0.01 mg/kg it was about 40 percent, and with a dose of 0.03 mg/kg it was 100 percent. For morphine (indicated by the red curve), duration of pain relief with a dose of 0.3 mg/kg was about 20 percent of the maximum possible effect, with a dose of 1 mg/kg it was about 50 percent, and with a dose of 2 mg/kg it was 100 percent.
Source:
- Ding, H., Kiguchi, N., Yasuda, D., et al. A bifunctional nociceptin and mu opioid receptor agonist is analgesic without opioid side effects in nonhuman primates. Science Translational Medicine 10(456): doi: 10.1126/scitranslmed.aar3483, 2018.